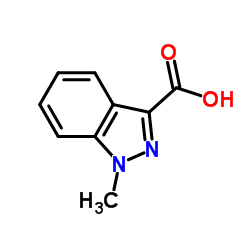
1-Methylindazole-3-carboxylic acid
- CAS No.50890-83-0
- GradeIndustrial / Pharmaceutical
- Availability● In Stock
High-purity pharmaceutical intermediate used primarily in the synthesis of Granisetron hydrochloride. Available in bulk with full quality documentation.
Request Bulk PricingProduct Technical Details
Product Overview
1-Methylindazole-3-carboxylic acid is a specialized organic compound widely recognized for its critical role in pharmaceutical synthesis. As a key building block, this chemical serves as an essential precursor in the manufacturing of advanced therapeutic agents, specifically within the antiemetic drug class. Our facility produces this intermediate under strict quality control protocols to ensure consistency and reliability for downstream pharmaceutical applications.
The compound is characterized by its stable indazole core structure, which provides the necessary chemical reactivity for subsequent coupling reactions. We understand the importance of molecular integrity in drug development, which is why our production process focuses on minimizing structural impurities and maximizing yield efficiency.
Key Specifications
| Parameter | Specification |
|---|---|
| CAS Number | 50890-83-0 |
| Molecular Formula | C9H8N2O2 |
| Molecular Weight | 176.17 g/mol |
| Appearance | White to off-white crystalline powder |
| Purity | ≥99.0% |
| Melting Point | 213~218°C |
| Loss on Drying | ≤0.5% |
| Total Impurities | ≤2.0% |
Industrial Applications
This chemical is predominantly utilized as a strategic intermediate in the synthesis of Granisetron hydrochloride, a potent serotonin 5-HT3 receptor antagonist. The high purity level of our 1-Methylindazole-3-carboxylic acid ensures that the final active pharmaceutical ingredient meets rigorous regulatory standards. Pharmaceutical manufacturers rely on consistent quality raw materials to maintain the safety and efficacy of their final formulations.
Beyond its primary use, this compound serves as a valuable reference standard in analytical chemistry, often identified as Granisetron Impurity D during quality control testing. This dual utility highlights its significance in both production and verification phases of the drug manufacturing lifecycle.
Quality Assurance
- Strict adherence to GMP guidelines during production.
- Comprehensive testing via HPLC and infrared spectroscopy.
- Certificate of Analysis (COA) provided with every batch.
- Low residual solvent content to ensure safety.
- Consistent batch-to-batch reproducibility.
Packaging and Storage
To maintain chemical stability, the product is packaged in 25kg cardboard drums or according to specific customer requirements. Each container is sealed to prevent moisture ingress and contamination. We recommend storing the material in a cool, dry, and well-closed container away from strong light and heat sources. Proper storage conditions are essential to preserve the physical and chemical properties of the compound over extended periods.
Our logistics team ensures secure global shipping, handling all documentation required for international chemical transport. For bulk orders or custom synthesis requests, our technical sales team is available to provide detailed specifications and competitive pricing structures tailored to your production needs.



