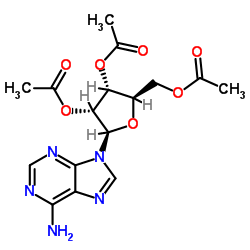
2',3',5'-Tri-O-acetyl-D-adenosine
- CAS No.7387-57-7
- GradeIndustrial / Pharmaceutical
- Availability● In Stock
High-purity 2',3',5'-Tri-O-acetyl-D-adenosine (CAS 7387-57-7) designed for pharmaceutical synthesis. Reliable supply with ≥98.0% assay specification for global research and production needs.
Request Bulk PricingProduct Technical Details
Product Overview
2',3',5'-Tri-O-acetyl-D-adenosine represents a critical protected nucleoside derivative extensively utilized in modern medicinal chemistry and organic synthesis. As a fully acetylated form of adenosine, this compound features acetyl groups protecting the hydroxyl functionalities at the 2', 3', and 5' positions of the ribose sugar moiety. This specific protection strategy is essential for preventing unwanted side reactions during complex multi-step synthesis pathways, particularly when constructing nucleotide analogs or modifying the nucleobase structure.
Our manufacturing facility produces this intermediate under strict quality control protocols to ensure consistent batch-to-batch reliability. The compound serves as a foundational building block for researchers and pharmaceutical companies developing antiviral agents, anticancer therapeutics, and various biochemical probes. The stability provided by the tri-O-acetyl protection allows chemists to perform selective transformations on the adenine base without compromising the integrity of the sugar backbone.
Technical Specifications
| Parameter | Specification |
|---|---|
| Chemical Name | 2',3',5'-Tri-O-acetyl-D-adenosine |
| CAS Number | 7387-57-7 |
| Molecular Formula | C16H19N5O7 |
| Molecular Weight | 393.35 g/mol |
| Appearance | White to off-white crystalline powder |
| Assay (HPLC) | ≥98.0% |
| Melting Point | 168-170°C |
| Density | 1.62 g/cm³ |
| Refractive Index | 1.679 |
Industrial Applications
The primary utility of 2',3',5'-Tri-O-acetyl-D-adenosine lies in its role as a versatile precursor for nucleoside analogue synthesis. In the pharmaceutical industry, protected nucleosides are indispensable for the development of next-generation antiviral medications. The acetyl groups can be selectively removed under mild basic conditions when the synthetic sequence requires exposure of the hydroxyl groups, offering significant flexibility in process chemistry.
- Synthesis of nucleotide prodrugs and antiviral active pharmaceutical ingredients.
- Development of anticancer agents targeting DNA and RNA synthesis pathways.
- Use as a standard reference material in analytical chemistry and quality control laboratories.
- Foundation for creating modified oligonucleotides used in genetic research and diagnostics.
Quality Assurance and Handling
We prioritize product integrity through comprehensive analytical testing. Every batch undergoes rigorous verification using High-Performance Liquid Chromatography (HPLC) to confirm purity levels meet or exceed the 98.0% threshold. Additionally, structural confirmation is supported by nuclear magnetic resonance (NMR) spectroscopy and mass spectrometry data available upon request. Our commitment to quality ensures that downstream synthesis processes proceed with minimal impurities, maximizing yield and reducing waste.
Regarding logistics, the product is packaged in 25 kg drums lined with moisture-proof materials to maintain stability during transit. Storage recommendations include keeping the container in a cool, ventilated area away from direct sunlight and strong oxidizing agents. Proper handling procedures should be followed to ensure safety, including the use of appropriate personal protective equipment. We provide full documentation, including Certificates of Analysis (COA), with every shipment to support regulatory compliance and quality assurance records for our global partners.



