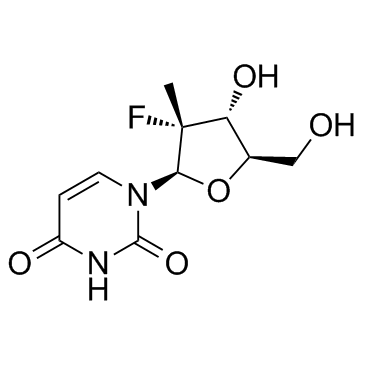
(2'R)-2'-Deoxy-2'-fluoro-2'-methyl-uridine
- CAS No.863329-66-2
- GradeIndustrial / Pharmaceutical
- Availability● In Stock
High-purity (2'R)-2'-Deoxy-2'-fluoro-2'-methyl-uridine, a key intermediate in antiviral drug synthesis, particularly for hepatitis C therapeutics.
Request Bulk PricingProduct Technical Details
Product Overview
(2'R)-2'-Deoxy-2'-fluoro-2'-methyl-uridine is a structurally modified nucleoside derivative widely employed as a critical pharmaceutical intermediate in the development of antiviral agents. This compound serves as the deaminated metabolite of β-D-2'-deoxy-2'-fluoro-2'-C-methylcytidine and plays a pivotal role in the synthetic pathway toward potent inhibitors of hepatitis C virus (HCV) replication. Its unique fluorinated and methylated sugar moiety enhances metabolic stability and binding affinity, making it indispensable in modern nucleotide analog synthesis.
Specifications
| Molecular Formula | C₁₀H₁₃FN₂O₅ |
|---|---|
| Molecular Weight | 260.22 g/mol |
| Melting Point | 237–238 °C |
| Density | 1.6 ± 0.1 g/cm³ |
| Refractive Index | 1.596 |
| Appearance | White to off-white powder |
| Assay (Purity) | ≥98.0% |
| Individual Impurity | ≤0.5% |
| Water Content | ≤0.5% |
| Loss on Drying | ≤1.0% |
| Residue on Ignition | ≤1.0% |
| Dichloromethane Residue | ≤600 ppm |
| Acetic Acid Residue | ≤5000 ppm |
| Methanol Residue | ≤3000 ppm |
Industrial Applications
This advanced nucleoside analog is primarily utilized as a building block in the multi-step synthesis of direct-acting antivirals (DAAs), notably serving as a precursor in the production of sofosbuvir-related intermediates (e.g., CAS 1190307-88-0). Its application is confined to research and large-scale pharmaceutical manufacturing under strict regulatory compliance. Key advantages include:
- High chemical and stereochemical purity ensuring reproducible reaction outcomes
- Optimized solubility profile for efficient downstream processing
- Compatibility with standard phosphoramidite and nucleotide coupling chemistries
- Rigorous quality control aligned with ICH guidelines for genotoxic impurities
Stored in tightly sealed containers under cool, dry, and well-ventilated conditions, this intermediate maintains stability during extended storage. Supplied in 25 kg fiber drums or custom packaging per client specifications, it supports seamless integration into GMP-compliant API manufacturing workflows.



