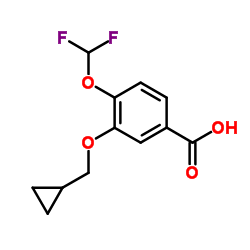
3-Cyclopropylmethoxy-4-difluoromethoxybenzoic Acid
- CAS No.162401-62-9
- GradeIndustrial / Pharmaceutical
- Availability● In Stock
High-purity 3-Cyclopropylmethoxy-4-difluoromethoxybenzoic Acid (CAS 162401-62-9) for pharmaceutical synthesis.
Request Bulk PricingProduct Technical Details
Product Overview
3-Cyclopropylmethoxy-4-difluoromethoxybenzoic Acid is a specialized organic compound serving as a critical building block in the pharmaceutical industry. Identified by CAS number 162401-62-9, this high-value intermediate plays an essential role in the synthesis of advanced therapeutic agents, particularly within the respiratory medicine sector. Our facility produces this compound under strict quality control protocols to ensure consistency and reliability for downstream manufacturing processes.
The molecular structure features both cyclopropylmethoxy and difluoromethoxy substituents on a benzoic acid backbone. This unique configuration imparts specific chemical properties required for complex multi-step synthesis routes. As a key precursor, it is primarily utilized in the production of Roflumilast-related pharmaceuticals, contributing to the development of treatments for chronic obstructive pulmonary disease (COPD).
Key Specifications
| Parameter | Value |
|---|---|
| CAS Number | 162401-62-9 |
| Molecular Formula | C12H12F2O4 |
| Molecular Weight | 258.218 |
| Appearance | White powder |
| Assay | ≥99.0% |
| Water Content | ≤0.50% |
| Density | 1.4±0.1 g/cm3 |
| Boiling Point | 356.4±37.0 °C at 760 mmHg |
| Flash Point | 169.4±26.5 °C |
Industrial Applications
This chemical intermediate is predominantly employed in the synthesis of phosphodiesterase-4 (PDE4) inhibitors. Its high purity level ensures minimal impurity carryover into final active pharmaceutical ingredients (APIs). Laboratories and manufacturing plants utilize this material for process development, scale-up studies, and commercial production of respiratory medications. The stability of the difluoromethoxy group under various reaction conditions makes it a preferred choice for medicinal chemists designing robust synthetic pathways.
Beyond its primary use, this compound serves as a reference standard for quality control laboratories analyzing Roflumilast impurities. Ensuring the availability of such high-grade intermediates supports regulatory compliance and accelerates the drug development timeline for pharmaceutical partners globally.
Quality Assurance and Packaging
We adhere to international quality management standards to guarantee product integrity. Each batch undergoes rigorous testing using advanced analytical techniques such as HPLC and NMR to verify identity and purity. A comprehensive Certificate of Analysis (COA) is provided with every shipment, detailing all critical quality attributes. Our packaging solutions are designed to maintain product stability during transit, typically supplied in 25kg drums or customized according to specific client requirements.
Storage and Handling
To preserve chemical stability, store this material in a cool, dry, and well-ventilated area. Keep containers tightly closed when not in use to prevent moisture absorption or contamination. Proper handling procedures should be followed in accordance with safety data sheets (SDS). Our technical support team is available to provide guidance on safe storage conditions and handling protocols to ensure workplace safety and product longevity.



