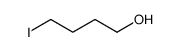
4-Iodo-1-butanol
- CAS No.3210-08-0
- GradeIndustrial / Pharmaceutical
- Availability● In Stock
High-purity 4-iodo-1-butanol (CAS 3210-08-0) is a versatile halogenated alcohol widely used as a building block in organic synthesis and pharmaceutical intermediate manufacturing.
Request Bulk PricingProduct Technical Details
Product Overview
4-Iodo-1-butanol (CAS 3210-08-0) is a colorless to pale yellow transparent liquid belonging to the class of iodinated aliphatic alcohols. With its bifunctional structure featuring both a primary hydroxyl group and a reactive iodine atom at opposite ends of a four-carbon chain, it serves as a highly valuable synthon in advanced organic chemistry. This compound is primarily employed as a key intermediate in the construction of complex molecular architectures, including heterocycles, polymers, and bioactive molecules.
Specifications
| Molecular Formula | C₄H₉IO |
|---|---|
| Molecular Weight | 200.02 g/mol |
| Assay | ≥90.0% |
| Moisture | ≤0.3% |
| Appearance | Colorless to yellow transparent liquid |
| Storage | Store in a cool, dry, and well-ventilated area away from light and incompatible materials |
| Packaging | 200 kg/drum; custom packaging available upon request |
Industrial Applications
As a specialty halogenated intermediate, 4-iodo-1-butanol plays a critical role in multi-step synthetic sequences. Its primary applications include:
- Serving as an alkylating agent in nucleophilic substitution reactions due to the excellent leaving-group ability of iodide
- Acting as a precursor for the preparation of cyclic ethers, lactones, and nitrogen-containing heterocycles
- Facilitating the synthesis of functionalized polymers and dendrimers through step-growth or click chemistry approaches
- Enabling the introduction of butanol-derived spacers in medicinal chemistry scaffolds and agrochemical intermediates
Manufacturers and R&D laboratories rely on high-purity 4-iodo-1-butanol to ensure reaction reproducibility, yield optimization, and minimal byproduct formation in sensitive transformations such as Williamson ether synthesis, Mitsunobu reactions, and transition-metal-catalyzed cross-couplings.



