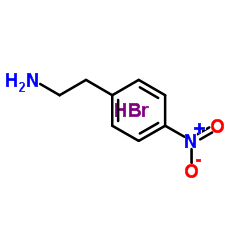
4-Nitrophenylethylamine Hydrobromide
- CAS No.69447-84-3
- GradeIndustrial / Pharmaceutical
- Availability● In Stock
High purity 4-Nitrophenylethylamine Hydrobromide designed for pharmaceutical synthesis. Reliable intermediate for complex organic manufacturing with full COA support.
Request Bulk PricingProduct Technical Details
Product Overview
4-Nitrophenylethylamine Hydrobromide is a specialized organic compound widely recognized for its stability and utility in medicinal chemistry. As a hydrobromide salt, this substance offers enhanced solubility and handling characteristics compared to its free base counterpart, making it an ideal candidate for multi-step synthesis protocols. Our manufacturing process ensures consistent molecular integrity, catering to the rigorous demands of pharmaceutical research and industrial production.
This chemical serves as a critical building block in the construction of complex therapeutic agents. The nitro group present in the structure provides a versatile handle for subsequent reduction or substitution reactions, enabling chemists to derive various amine functionalities required in advanced drug discovery pipelines. We prioritize batch-to-batch consistency to support scalable manufacturing operations.
Technical Specifications
| Parameter | Specification |
|---|---|
| CAS Number | 69447-84-3 |
| Molecular Formula | C8H11BrN2O2 |
| Molecular Weight | 247.089 g/mol |
| Appearance | Off-white to light yellow crystalline powder |
| Assay | ≥98.0% |
| Boiling Point | 340.9°C at 760 mmHg |
| Flash Point | 159.9°C |
Industrial Applications
Primarily utilized as a key intermediate in the synthesis of cardiovascular pharmaceuticals, this compound plays a vital role in the production pathway of antiarrhythmic agents. Its high purity profile minimizes impurity carryover in downstream reactions, which is essential for meeting regulatory standards in active pharmaceutical ingredient manufacturing. Researchers value this material for its predictable reactivity during nucleophilic substitution and coupling processes.
Beyond specific drug synthesis, it functions as a valuable reference standard in analytical laboratories. The stable crystalline nature allows for precise weighing and solution preparation, facilitating accurate kinetic studies and method validation. Our supply chain supports both pilot-scale research and large-volume commercial production requirements.
Quality Assurance and Logistics
Quality control is paramount in our production facility. Each batch undergoes comprehensive testing using high-performance liquid chromatography and nuclear magnetic resonance spectroscopy to verify structural identity and purity levels. We provide a Certificate of Analysis with every shipment, ensuring full transparency and compliance with international chemical safety standards.
- Packaged in 25 kg drums for optimal protection during transit.
- Custom packaging solutions available upon client request.
- Stored in cool, ventilated environments to maintain stability.
- Global shipping capabilities with secure hazardous material handling.
We are committed to delivering reliable chemical solutions with timely logistics support. Our technical team is available to assist with synthesis route optimization and safety data inquiries, ensuring a seamless integration into your manufacturing workflow.



