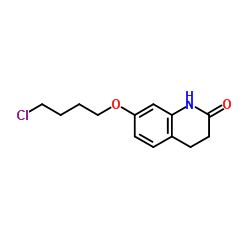
7-(4-Chlorobutoxy)-3,4-dihydroquinolin-2(1H)-one
- CAS No.120004-79-7
- GradeIndustrial / Pharmaceutical
- Availability● In Stock
High purity 7-(4-Chlorobutoxy)-3,4-dihydroquinolin-2(1H)-one designed for pharmaceutical synthesis. Reliable bulk supply with full documentation for global manufacturing partners.
Request Bulk PricingProduct Technical Details
Product Overview
7-(4-Chlorobutoxy)-3,4-dihydroquinolin-2(1H)-one is a specialized organic compound serving as a critical building block in the pharmaceutical industry. This quinolinone derivative is meticulously synthesized to meet stringent quality standards required for advanced medicinal chemistry. Our production facilities adhere to rigorous protocols to ensure consistency, purity, and reliability for downstream applications.
As a key precursor, this compound plays an integral role in the synthesis of complex therapeutic agents. The chemical structure features a chlorobutoxy side chain attached to a dihydroquinolinone core, providing specific reactivity profiles essential for multi-step organic synthesis. We prioritize safety and quality throughout the manufacturing lifecycle.
Technical Specifications
| Parameter | Specification |
|---|---|
| Chemical Name | 7-(4-Chlorobutoxy)-3,4-dihydroquinolin-2(1H)-one |
| CAS Number | 120004-79-7 |
| Molecular Formula | C13H16ClNO2 |
| Molecular Weight | 253.73 g/mol |
| Appearance | White to off-white crystalline powder |
| Purity | ≥98% |
| Density | 1.187 g/cm3 |
| Boiling Point | 448.925°C at 760 mmHg |
| Flash Point | 225.302°C |
Industrial Applications
This intermediate is primarily utilized in the manufacture of pharmaceutical APIs, specifically within the class of antipsychotic therapeutics. It serves as a vital coupling component in the construction of complex molecular architectures. Chemical manufacturers and research institutions rely on this material for process development and commercial-scale production.
Our supply chain supports various stages of drug development, from early-stage research to full-scale commercialization. The high purity level ensures minimal impurity carryover into final products, reducing downstream purification burdens and enhancing overall process efficiency.
Quality Assurance and Storage
Every batch undergoes comprehensive analytical testing, including HPLC, NMR, and mass spectrometry, to verify identity and purity. We provide a Certificate of Analysis (COA) with each shipment to guarantee compliance with specified parameters. Our quality management system is designed to maintain product integrity from synthesis to delivery.
For optimal stability, store this compound in a cool, dry, and well-ventilated area. Keep containers tightly closed to protect from moisture and strong light. Avoid exposure to extreme temperatures or incompatible chemical environments. Proper handling procedures should be followed in accordance with local safety regulations and material safety data sheets.
Why Choose Our Supply
- Consistent high purity levels suitable for sensitive synthetic routes
- Scalable production capacity to meet bulk demand fluctuations
- Comprehensive technical support and documentation package
- Global logistics network ensuring timely and secure delivery
- Competitive pricing structures for long-term partnerships
We are committed to supporting the pharmaceutical industry with reliable chemical solutions. Our team of experts is available to discuss custom synthesis requirements or provide additional technical data to facilitate your production processes.



