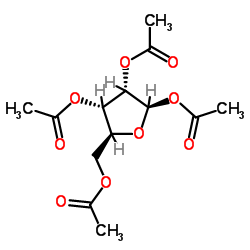
Beta-D-Ribofuranose 1,2,3,5-Tetraacetate
- CAS No.13035-61-5
- GradeIndustrial / Pharmaceutical
- Availability● In Stock
High purity Beta-D-Ribofuranose 1,2,3,5-Tetraacetate is a critical protected sugar intermediate used extensively in the synthesis of nucleoside analogs and pharmaceutical APIs.
Request Bulk PricingProduct Technical Details
Product Overview
Beta-D-Ribofuranose 1,2,3,5-Tetraacetate is a highly specialized carbohydrate derivative designed for advanced pharmaceutical synthesis. As a fully acetylated protected form of ribose, this compound serves as a fundamental building block in the construction of complex nucleoside structures. The acetylation at the 1, 2, 3, and 5 positions provides exceptional stability during glycosylation reactions, ensuring high stereochemical control in downstream processes. Our manufacturing process adheres to strict quality control protocols to deliver material suitable for research and development of novel therapeutic agents.
In the realm of medicinal chemistry, protected sugars are indispensable for the creation of nucleoside analogs, which are pivotal in developing antiviral and anticancer medications. This tetraacetate variant offers superior solubility in organic solvents compared to unprotected sugars, facilitating smoother reaction kinetics and easier purification steps. We supply this intermediate with a focus on consistency, batch-to-batch reproducibility, and comprehensive documentation to support regulatory filings.
Key Specifications
| Parameter | Specification |
|---|---|
| Appearance | White to off-white crystalline powder |
| CAS Number | 13035-61-5 |
| Molecular Formula | C13H18O9 |
| Molecular Weight | 318.28 g/mol |
| Assay (HPLC) | ≥98.0% |
| Melting Point | 81-83°C |
| Loss on Drying | ≤0.5% |
| Residue on Ignition | ≤0.1% |
| Heavy Metals | ≤20 ppm |
Industrial Applications
This compound is primarily utilized as a key intermediate in the synthesis of nucleoside analogs. It acts as a glycosyl donor in coupling reactions with heterocyclic bases, enabling the formation of beta-configured nucleosides. The high purity level ensures minimal side reactions, which is critical when synthesizing complex molecules where impurity profiles must be tightly controlled. Common applications include the production of uridine derivatives and other pyrimidine nucleoside analogs used in pharmaceutical research.
Beyond direct synthesis, this material is valuable for process development teams optimizing glycosylation routes. The predictable reactivity of the tetraacetate group allows chemists to streamline scale-up processes from laboratory to pilot plant. Our supply chain supports both small-scale research requirements and large-scale commercial manufacturing needs, providing flexibility for clients at various stages of drug development.
Quality Assurance and Packaging
Quality is paramount in pharmaceutical intermediates. Each batch undergoes rigorous testing using high-performance liquid chromatography (HPLC) and nuclear magnetic resonance (NMR) to confirm identity and purity. We provide a comprehensive Certificate of Analysis (COA) with every shipment, detailing all critical quality attributes. Our facility maintains strict environmental controls to prevent moisture uptake, ensuring the product remains stable during storage.
- Consistent high purity levels exceeding 98 percent
- Stable supply chain with global shipping capabilities
- Custom packaging options available upon request
- Technical support for synthesis optimization
- Compliance with international safety and quality standards
Standard packaging consists of 25 kg drums, lined with moisture-proof bags to maintain integrity. For specialized requirements, we offer flexible packaging solutions tailored to customer specifications. Storage recommendations include keeping the material in a cool, ventilated area away from direct sunlight and strong oxidizing agents. Proper handling ensures the longevity and performance of this critical chemical intermediate in your synthesis workflows.



