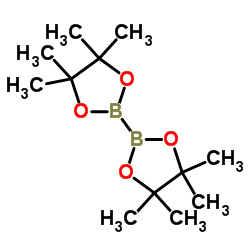
Bis(pinacolato)diboron
- CAS No.73183-34-3
- GradeIndustrial / Pharmaceutical
- Availability● In Stock
High-purity Bis(pinacolato)diboron designed for advanced organic synthesis and pharmaceutical research applications.
Request Bulk PricingProduct Technical Details
Product Overview
Bis(pinacolato)diboron, frequently abbreviated as BPDB, represents a cornerstone reagent in modern organic synthesis and medicinal chemistry. As a stable organoboron compound, it offers significant advantages over traditional boronic acids regarding handling, storage, and reactivity. Our manufacturing process ensures exceptional purity levels, making it an ideal choice for complex molecular constructions where precision is paramount.
This compound features two pinacol boronate ester groups attached to a central boron-boron bond. This unique structure provides enhanced stability against protodeboronation and oxidation, allowing chemists to perform reactions under milder conditions. It is widely recognized as a superior substrate for transitioning metal-catalyzed cross-coupling reactions, particularly in the development of advanced pharmaceutical intermediates and functional materials.
Key Specifications
| Parameter | Value |
|---|---|
| Product Name | Bis(pinacolato)diboron |
| CAS Number | 73183-34-3 |
| Molecular Formula | C12H24B2O4 |
| Molecular Weight | 253.94 g/mol |
| Appearance | White powder |
| Purity (Assay) | ≥99.0% |
| Water Content | ≤0.50% |
| Melting Point | 137-140 °C |
| Boiling Point | 222.6 °C at 760 mmHg |
| Density | 0.97 g/cm3 |
Industrial Applications
The versatility of Bis(pinacolato)diboron makes it indispensable in various high-value chemical sectors. Its primary utility lies in its ability to introduce boron functionality into organic molecules efficiently. Key application areas include:
- Suzuki-Miyaura Cross-Coupling: Serving as a critical boration reagent for coupling aromatic and vinyl halides.
- Medicinal Chemistry: Facilitating the synthesis of complex drug candidates and therapeutic intermediates.
- C-H Activation: Enabling direct functionalization of unactivated carbon-hydrogen bonds.
- Material Science: Used in the preparation of organic electronic materials and polymers.
- Palladium Catalysis: Acting as a substrate in cyclization reactions involving enynes.
Quality Assurance and Safety
We adhere to strict quality control protocols to ensure every batch meets international standards. Each shipment is accompanied by a comprehensive Certificate of Analysis (COA) verifying identity and purity. Regarding safety, while classified as non-dangerous goods for transport, proper handling procedures are essential. The product should be stored in a cool, dry, and well-ventilated area with containers tightly closed to prevent moisture absorption. We recommend using inert atmosphere techniques during handling to maintain optimal reactivity.
Our global supply chain ensures reliable delivery for research and industrial scales. Whether for pilot plant operations or large-scale manufacturing, our team provides technical support to optimize your synthesis routes. Contact us today for detailed specifications, bulk pricing, and custom packaging solutions tailored to your operational requirements.



