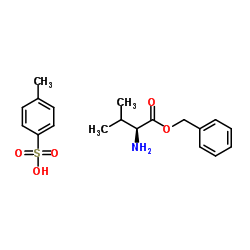
L-Valine Benzyl Ester p-Toluenesulfonate Salt
- CAS No.16652-76-9
- GradeIndustrial / Pharmaceutical
- Availability● In Stock
High-purity L-Valine benzyl ester p-toluenesulfonate salt designed for Valsartan intermediate synthesis. Rigorous quality control ensures optimal reaction performance.
Request Bulk PricingProduct Technical Details
Product Overview
L-Valine benzyl ester p-toluenesulfonate salt is a critical chiral building block extensively utilized in the pharmaceutical industry, specifically for the synthesis of Valsartan, a widely prescribed antihypertensive agent. This compound serves as a key intermediate, providing the necessary stereochemical configuration required for effective API manufacturing. Our facility produces this material under strict GMP-like conditions to ensure consistency and reliability for downstream processes.
The p-toluenesulfonate salt form enhances the stability and solubility of the amino ester during organic synthesis, facilitating smoother reaction kinetics in peptide coupling and alkylation steps. We understand the importance of optical purity in pharmaceutical applications, which is why our production process emphasizes the retention of the L-configuration throughout crystallization and drying phases.
Technical Specifications
| Parameter | Specification |
|---|---|
| CAS Number | 16652-76-9 |
| Molecular Formula | C19H25NO5S |
| Molecular Weight | 379.47 g/mol |
| Appearance | White Crystal |
| Assay (HPLC) | ≥99.0% |
| Specific Rotation | -3.8° to -3.2° |
| Melting Point | 155-162°C |
| Loss on Drying | ≤1.0% |
| Residue on Ignition | ≤0.1% |
| Single Impurity | ≤0.5% |
Quality Assurance and Control
Quality is paramount in the production of pharmaceutical intermediates. Each batch of L-Valine benzyl ester p-toluenesulfonate salt undergoes comprehensive analytical testing. We employ high-performance liquid chromatography (HPLC) to verify assay purity and monitor potential impurities such as L-Leucine, L-Isoleucine, and L-Alanine derivatives, ensuring they remain below specified thresholds. Optical rotation is measured to confirm chiral integrity, which is vital for the biological activity of the final drug product.
Our quality management system ensures full traceability from raw material sourcing to final packaging. Certificates of Analysis (COA) are provided with every shipment, detailing all test results against internal and pharmacopoeial standards. This transparency allows our partners to validate materials quickly upon receipt, minimizing downtime in their production schedules.
Storage and Handling
To maintain product stability, this chemical should be stored in a cool, ventilated area away from direct sunlight and moisture. The recommended storage temperature is between 2-8°C for long-term preservation, although ambient storage is acceptable for short durations if sealed properly. Containers must be kept tightly closed to prevent hydrolysis of the ester group. Standard laboratory safety protocols should be followed during handling, including the use of appropriate personal protective equipment.
Industrial Applications
- Primary intermediate for the commercial synthesis of Valsartan.
- Chiral synthon for peptide chemistry and medicinal research.
- Building block for developing angiotensin II receptor antagonists.
- Reference standard for analytical method validation in QC laboratories.
We offer flexible packaging options, including 25 kg drums, to accommodate both pilot-scale and large-scale manufacturing needs. Our global logistics network ensures timely delivery to pharmaceutical manufacturers worldwide, supported by technical documentation for regulatory submissions.



