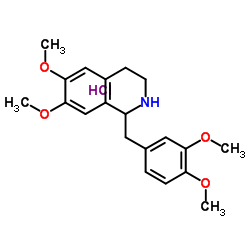
(R)-1,2,3,4-Tetrahydropapaverine Hydrochloride
- CAS No.54417-53-7
- GradeIndustrial / Pharmaceutical
- Availability● In Stock
High-purity pharmaceutical intermediate used in the synthesis of neuromuscular blocking agents. Available in bulk with comprehensive COA.
Request Bulk PricingProduct Technical Details
Product Overview
(R)-1,2,3,4-Tetrahydropapaverine Hydrochloride is a specialized chiral pharmaceutical intermediate designed for high-level organic synthesis within the medicinal chemistry sector. This compound serves as a critical building block in the manufacturing pipeline of advanced neuromuscular blocking agents, specifically acting as a key precursor for Atracurium Besilate. Our facility ensures that every batch meets stringent enantiomeric purity standards, which is essential for maintaining the efficacy and safety profile of the final active pharmaceutical ingredient.
As a dedicated manufacturer with decades of experience in complex organic synthesis, we understand the vital role this intermediate plays in the production of muscle relaxants used in surgical anesthesia. The precise stereochemistry of the R-enantiomer is maintained through rigorous process control, ensuring consistent performance in downstream reactions. We commit to supplying global pharmaceutical partners with material that adheres to international quality benchmarks.
Technical Specifications
| Property | Specification |
|---|---|
| CAS Number | 54417-53-7 |
| Molecular Formula | C20H26ClNO4 |
| Molecular Weight | 379.878 g/mol |
| Appearance | White to off-white powder |
| Assay (Purity) | ≥99.0% |
| Density | 1.12 g/cm3 |
| Boiling Point | 475.8ºC at 760 mmHg |
| Flash Point | 202.7ºC |
| Refractive Index | 1.549 |
Industrial Applications
The primary application of this high-grade intermediate lies within the pharmaceutical industry, specifically for the synthesis of non-depolarizing neuromuscular blocking drugs. Its structural integrity allows for efficient coupling reactions required to produce agents that facilitate endotracheal intubation and skeletal muscle relaxation during surgery. Beyond this specific use, the compound serves as a valuable reference standard and research material for laboratories focusing on isoquinoline alkaloid derivatives and chiral synthesis methodologies.
Our production process is optimized to minimize impurities that could affect downstream catalytic steps. We work closely with process chemists to ensure that the physical properties, such as particle size and solubility profiles, are consistent with the requirements of large-scale reactor setups. This reliability helps our clients reduce waste and improve overall yield in their final API manufacturing processes.
Quality Assurance and Storage
Quality is the cornerstone of our manufacturing philosophy. Each production lot undergoes comprehensive analytical testing, including HPLC, NMR, and mass spectrometry, to verify chemical identity and purity levels. A detailed Certificate of Analysis (COA) is provided with every shipment, documenting all critical quality attributes. We maintain full traceability from raw material sourcing to final packaging.
To preserve stability, the product should be stored in a cool, ventilated environment away from direct sunlight and moisture. Standard packaging includes 25 kg drums, but we offer flexible custom packaging solutions to meet specific logistical needs. Our global supply chain ensures timely delivery while maintaining the integrity of the chemical substance throughout transit. For bulk inquiries or technical support regarding synthesis routes, our expert team is available to assist.



