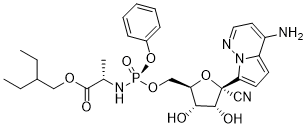
Remdesivir
- CAS No.1809249-37-3
- GradeIndustrial / Pharmaceutical
- Availability● In Stock
High purity Remdesivir API (CAS 1809249-37-3) for pharmaceutical research and manufacturing. Compliant with strict quality standards, available for global bulk supply with full documentation.
Request Bulk PricingProduct Technical Details
Product Overview
Remdesivir, also known by its development code GS-5734, is a sophisticated nucleotide analog pharmaceutical ingredient designed for advanced antiviral research and therapeutic development. As a prodrug of the active metabolite GS-441524, this compound represents a critical building block in the synthesis of broad-spectrum antiviral medications. Our facility specializes in the production of high-quality Remdesivir intermediates and API grades, ensuring consistency and reliability for pharmaceutical manufacturers and research institutions worldwide.
We understand the critical nature of supply chain stability in the pharmaceutical industry. Therefore, our manufacturing processes are optimized to deliver consistent batches that meet rigorous international standards. This product is intended for professional use in licensed pharmaceutical formulation, clinical research, and chemical development projects.
Chemical Properties and Structure
Chemically, Remdesivir is characterized as a complex organic phosphate ester. The molecular formula C28H36N5O8P reflects its intricate structure, which includes a modified nucleoside base linked to a phosphoramidate moiety. This specific configuration enhances cellular uptake and metabolic stability, making it a highly effective candidate for antiviral applications. The compound exhibits specific stereochemistry, denoted by the (S) and (2R,3S,4R,5R) configurations in its IUPAC name, which are critical for its biological activity.
The substance typically presents as a white to off-white crystalline powder. It is insoluble in water but demonstrates solubility in organic solvents such as ethanol, which facilitates various processing and formulation techniques. The specific rotation values are tightly controlled to ensure enantiomeric purity, a key factor in maintaining the efficacy and safety profile of the final pharmaceutical product.
Quality Specifications and Analysis
Quality assurance is paramount in our production workflow. Each batch undergoes comprehensive testing using high-performance liquid chromatography (HPLC) and other analytical methods to verify identity, purity, and impurity profiles. We maintain strict controls over residual solvents, heavy metals, and related substances to ensure compliance with pharmacopeial standards.
| Items of Analysis | Standard | Test Results |
|---|---|---|
| Appearance | White to off-white powder | White powder |
| Identification (HPLC) | Consistent with standard | Conforms |
| Solubility | Insoluble in water, soluble in ethanol | Conforms |
| Water Content | ≤1.0% | 0.07% |
| Specific Rotation | -18° to -25° | -22.63° |
| Heavy Metals | ≤10ppm | Conforms |
| Related Substances | Total impurities ≤0.3% | 0.16% |
| Purity (HPLC) | ≥99.7% | 99.84% |
| Assay (HPLC) | 98.0% to 102.0% | 99.7% |
Industrial Applications
This chemical is primarily utilized in the pharmaceutical sector for the development of antiviral therapies. It serves as a key active pharmaceutical ingredient (API) or a advanced intermediate in the synthesis of medications targeting RNA polymerase. Research applications include investigating efficacy against various viral pathogens, including coronaviruses and other RNA viruses. Additionally, it is used in formulation studies to optimize drug delivery systems and stability profiles.
Our clients leverage this material for creating drop-in replacements in existing development pipelines, establishing performance benchmarks for new formulations, and conducting equivalence studies. The high purity level ensures minimal interference in downstream processing, reducing the need for extensive purification steps during drug manufacturing.
Storage and Handling
To maintain stability and integrity, Remdesivir should be stored in a cool, dry place away from direct light and moisture. Containers must be kept tightly sealed when not in use to prevent degradation. Handling should be performed in accordance with standard laboratory safety protocols, including the use of appropriate personal protective equipment. We provide comprehensive documentation, including Certificates of Analysis (COA), to support regulatory filings and quality audits.



