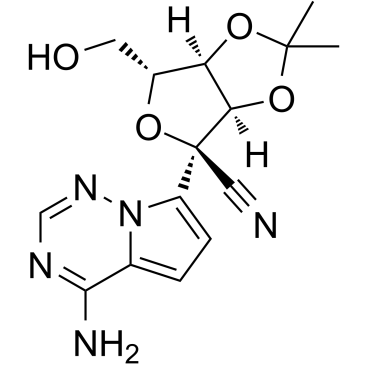
Remdesivir O-Desphosphate Acetonide Impurity
- CAS No.1191237-80-5
- GradeIndustrial / Pharmaceutical
- Availability● In Stock
High purity Remdesivir O-Desphosphate Acetonide Impurity (CAS 1191237-80-5) designed for pharmaceutical research and analytical method validation. Certified quality with comprehensive COA.
Request Bulk PricingProduct Technical Details
Product Overview
Our Remdesivir O-Desphosphate Acetonide Impurity is a critical reference standard and intermediate utilized extensively in the pharmaceutical research and development sector. This compound serves as a key structural analog associated with the synthesis pathway of Remdesivir, a notable nucleoside analog investigated for antiviral applications. As a specialized chemical intermediate, it plays an indispensable role in impurity profiling, method validation, and quality control processes within regulated laboratory environments.
We understand the stringent requirements of modern pharmaceutical analysis. Therefore, this product is manufactured under strict quality management systems to ensure consistency and reliability. It is intended strictly for research and development use by qualified professionals in industrial or academic settings. Our commitment to excellence ensures that every batch meets the highest standards of chemical purity and structural integrity required for precise analytical outcomes.
Chemical Properties And Structure
The chemical structure of this impurity features a complex heterocyclic system characterized by a pyrrolo-triazine core linked to a modified sugar moiety. Specifically, the molecule contains an acetonide protecting group and a nitrile functionality, which are crucial for its stability and reactivity during synthetic transformations. The stereochemistry is meticulously controlled, with specific configurations at multiple chiral centers ensuring that the material accurately reflects the target impurity profile found in final drug substances.
Understanding the physicochemical properties of this intermediate is vital for researchers developing robust analytical methods. The compound exhibits specific solubility characteristics and stability profiles that must be accounted for during storage and handling. Proper handling ensures that the material remains stable over time, preserving its utility as a reliable reference standard for high-performance liquid chromatography (HPLC) and other spectroscopic techniques.
Quality Specifications
Quality assurance is the cornerstone of our manufacturing process. Each batch undergoes rigorous testing using advanced analytical instrumentation to verify identity, purity, and stereochemical composition. We adhere to international guidelines for impurity standards, ensuring that our products support regulatory submissions and internal quality audits. The following table outlines the typical specifications for this material:
| Items of Analysis | Standard of Analysis | Test Results |
|---|---|---|
| Appearance | White solid | Conforms |
| Identification | HPLC | Conforms |
| Purity (HPLC) | ≥98.0% | 99.6% |
| E.e | ≥98.0% | 99.2% |
| Conclusion | Conforms to Factory Standard | |
Industrial Applications
This intermediate is primarily utilized in the development and validation of analytical methods for antiviral pharmaceuticals. Researchers employ this material to spike samples, calibrate instruments, and establish detection limits for impurity testing. Additionally, it serves as a valuable tool for studying degradation pathways and stability indicating methods. By utilizing high-quality impurity standards, pharmaceutical companies can ensure the safety and efficacy of their final products through comprehensive quality control.
- Impurity profiling and identification in drug substance analysis.
- Validation of chromatographic methods for regulatory compliance.
- Research into synthetic routes and process optimization.
- Stability studies and degradation product characterization.
Packaging And Storage
To maintain the integrity of the chemical structure, this product is packaged in sealed containers under inert atmosphere conditions when necessary. We recommend storing the material in a cool, dry place away from direct light and moisture. Proper storage conditions are essential to prevent hydrolysis or other degradation reactions that could compromise the purity of the standard. Each shipment includes a comprehensive Certificate of Analysis (COA) detailing the specific test results for that batch, ensuring full traceability and transparency for our global clients.



