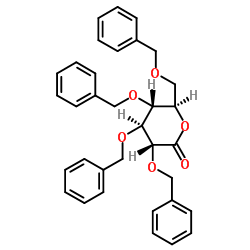
2,3,4,6-Tetra-O-benzyl-D-gluconic acid-δ-lactone
- CAS No.13096-62-3
- GradeIndustrial / Pharmaceutical
- Availability● In Stock
High-purity 2,3,4,6-Tetra-O-benzyl-D-gluconic acid-δ-lactone (CAS 13096-62-3) serves as a critical intermediate for SGLT2 inhibitor synthesis. Available in bulk with full COA.
Request Bulk PricingProduct Technical Details
Product Overview
2,3,4,6-Tetra-O-benzyl-D-gluconic acid-δ-lactone is a highly specialized pharmaceutical intermediate essential for the synthesis of next-generation antidiabetic medications. This benzyl-protected gluconolactone derivative plays a pivotal role in the construction of sodium-glucose cotransporter 2 (SGLT2) inhibitors. Our manufacturing process ensures exceptional chemical stability and stereochemical integrity, making it an ideal building block for complex organic synthesis in the pharmaceutical sector.
As a key precursor, this compound facilitates the efficient production of leading diabetes therapeutics. We adhere to strict quality control protocols to guarantee consistency across batches, supporting both research and large-scale commercial manufacturing needs.
Specifications and Chemical Properties
| Parameter | Specification |
|---|---|
| Chemical Name | 2,3,4,6-Tetra-O-benzyl-D-gluconic acid-δ-lactone |
| CAS Number | 13096-62-3 |
| Molecular Formula | C34H34O6 |
| Molecular Weight | 538.63 g/mol |
| Appearance | Light yellow liquid or crystal |
| Purity | ≥95.0% (HPLC) |
| Storage Conditions | Store in a cool, ventilated place |
Industrial Applications
This compound is primarily utilized as a critical intermediate in the manufacturing of SGLT2 inhibitors, a class of drugs used to manage type 2 diabetes. Specific applications include the synthesis of active pharmaceutical ingredients (APIs) such as Ipragliflozin, Empagliflozin, Dapagliflozin, and Tofogliflozin. The benzyl protection groups on the gluconic acid backbone allow for selective chemical transformations required during the multi-step synthesis of these high-value therapeutics.
Our product supports pharmaceutical companies and contract research organizations in developing robust synthesis routes. The high purity level minimizes downstream purification challenges, enhancing overall process efficiency and yield.
Quality Assurance and Packaging
Quality is paramount in pharmaceutical intermediate supply. Each batch undergoes rigorous testing using advanced analytical techniques, including HPLC and NMR, to verify identity and purity. We provide a comprehensive Certificate of Analysis (COA) with every shipment to ensure compliance with your internal quality standards.
- Consistent batch-to-batch reproducibility
- Comprehensive documentation and regulatory support
- Flexible packaging options to suit production scales
Standard packaging is available in 25 kg drums, though we offer customized packaging solutions based on specific customer requirements and logistics needs. Proper storage in a cool, ventilated environment is recommended to maintain product stability over time.
Regulatory Compliance
We are committed to full regulatory compliance and intellectual property respect. Please note that products covered by valid patents will not be offered to countries where such patents are in force. We encourage all clients to verify local regulatory requirements before procurement. Our team is available to provide technical support and documentation to facilitate your compliance processes.



