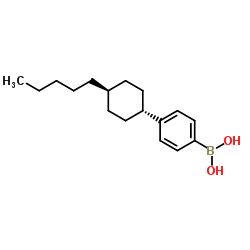
4-(Trans-4-pentylcyclohexyl)phenylboronic Acid
- CAS No.143651-26-7
- GradeIndustrial / Pharmaceutical
- Availability● In Stock
High purity 4-(Trans-4-pentylcyclohexyl)phenylboronic Acid for pharmaceutical synthesis. Reliable bulk supply with COA.
Request Bulk PricingProduct Technical Details
Product Overview
4-(Trans-4-pentylcyclohexyl)phenylboronic Acid is a specialized organic boron compound designed for advanced pharmaceutical and chemical synthesis applications. As a key aryl boronic acid derivative, this substance serves as a critical building block in the construction of complex molecular architectures, particularly within the realm of medicinal chemistry and material science. Our manufacturing process ensures exceptional consistency and purity, making it an ideal choice for research and development teams requiring reliable reagents for cross-coupling reactions.
This compound features a stable cyclohexyl structure linked to a phenyl boronic acid moiety, providing unique steric and electronic properties that enhance reaction efficiency. The trans-configuration of the pentylcyclohexyl group contributes to its specific reactivity profile, allowing for precise control during synthetic transformations. We adhere to strict quality management systems to guarantee that every batch meets rigorous international standards for chemical intermediates.
Key Specifications
| Parameter | Specification |
|---|---|
| CAS Number | 143651-26-7 |
| Molecular Formula | C17H27BO2 |
| Molecular Weight | 274.21 g/mol |
| Appearance | White powder |
| Assay (Purity) | ≥98.0% |
| Water Content | ≤0.50% |
| Density | 1.0±0.1 g/cm3 |
| Boiling Point | 421.1±38.0 °C at 760 mmHg |
| Flash Point | 208.5±26.8 °C |
Applications & Benefits
This boronic acid derivative is primarily utilized as a versatile intermediate in the synthesis of active pharmaceutical ingredients (APIs) and fine chemicals. Its primary application lies in Suzuki-Miyaura cross-coupling reactions, where it acts as a nucleophilic partner to form carbon-carbon bonds with high selectivity. The robust nature of the cyclohexyl ring enhances the stability of the molecule during various synthetic steps, reducing side reactions and improving overall yield.
- High reactivity in palladium-catalyzed coupling processes.
- Excellent solubility profile compatible with common organic solvents.
- Stable storage characteristics under recommended conditions.
- Supports the development of novel therapeutic compounds.
- Validated for use in scale-up manufacturing environments.
Storage & Handling
To maintain optimal quality, 4-(Trans-4-pentylcyclohexyl)phenylboronic Acid should be stored in a cool, dry, and well-ventilated area. Containers must remain tightly closed to prevent exposure to moisture and air, which could potentially affect the boronic acid functionality over extended periods. The product is classified under UN3277 for transport, requiring adherence to specific safety guidelines regarding hazardous materials. Personnel handling this chemical should utilize appropriate personal protective equipment, including gloves and eye protection, to ensure safety during weighing and transfer operations. For large-scale industrial use, we provide comprehensive technical data sheets and safety documentation to support compliant handling procedures.



