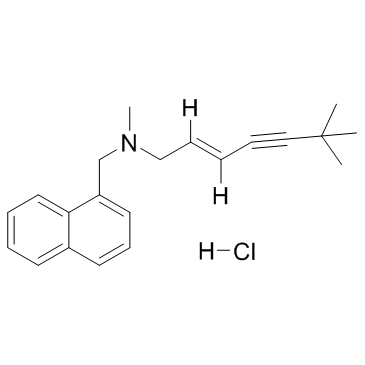
Terbinafine Hydrochloride
- CAS No.78628-80-5
- GradeIndustrial / Pharmaceutical
- Availability● In Stock
High purity Terbinafine Hydrochloride API for pharmaceutical formulations. Certified quality with comprehensive COA and global bulk supply capabilities.
Request Bulk PricingProduct Technical Details
Product Overview
Terbinafine Hydrochloride is a highly effective allylamine antifungal agent widely utilized in the pharmaceutical industry for the treatment of various fungal infections. As a critical active pharmaceutical ingredient (API), it serves as the foundational component for numerous dermatological formulations, including topical creams, gels, and oral tablets. Our manufacturing process ensures exceptional chemical stability and bioavailability, making it an ideal choice for pharmaceutical companies seeking reliable raw materials for antifungal therapies.
Chemically known as (E)-N-(6,6-Dimethyl-2-hepten-4-ynyl)-N-methyl-1-naphthalenemethanamine monohydrochloride, this compound exhibits superior solubility characteristics and potency. We adhere to strict international quality standards to guarantee that every batch meets the rigorous demands of modern medicine production. Our facility is equipped with advanced analytical instrumentation to verify identity and purity before shipment.
Mechanism of Action
The therapeutic efficacy of Terbinafine Hydrochloride stems from its specific inhibition of squalene epoxidase, a key enzyme in the fungal ergosterol biosynthesis pathway. By blocking this enzyme, the compound causes a lethal accumulation of squalene within the fungal cell while simultaneously depriving the cell of ergosterol, which is essential for membrane integrity. This dual action results in potent fungicidal activity against a broad spectrum of dermatophytes and yeasts, including Candida species. The Ki value of approximately 30 nM underscores its high affinity and effectiveness as a non-competitive inhibitor.
Quality Control and Specifications
Quality assurance is paramount in the production of pharmaceutical intermediates and APIs. Our comprehensive quality control protocol employs high-performance liquid chromatography (HPLC) and nuclear magnetic resonance (NMR) spectroscopy to validate chemical identity and purity levels. Each production lot undergoes meticulous testing to ensure compliance with factory standards and client specifications. We provide a detailed Certificate of Analysis (COA) with every order, documenting critical parameters such as melting point, loss on drying, and assay results.
| Items of Analysis | Standard of Analysis | Test Results |
|---|---|---|
| Appearance | White or off-white powder | Off-white powder |
| Identity by 1H-NMR | Reference to original data | Conform |
| Purity by HPLC | ≥94.0% (Area nominalization) | 94.15% |
| Melting Point | For information | 200.2~200.6 °C |
| Loss on drying | ≤0.50% (w/w) | 0.46% |
| Conclusion | Conforms to Factory Standard | |
Storage and Handling
To maintain optimal stability and potency, Terbinafine Hydrochloride should be stored in a cool, dry environment away from direct light and moisture. While the compound is relatively stable under ambient conditions, long-term storage at controlled temperatures such as -20°C is recommended for extended shelf life. Proper sealing of containers is essential to prevent hygroscopic absorption, which could affect flowability and processing characteristics during formulation. Always handle with appropriate personal protective equipment to ensure safety in the laboratory or manufacturing setting.
Industrial Applications
This material is primarily destined for the manufacture of antifungal medications used in human healthcare. It is compatible with various excipients and binding agents commonly used in tablet compression and topical ointment preparation. Pharmaceutical formulators value this ingredient for its consistent performance profile and well-documented safety record. Whether developing generic equivalents or new dermatological solutions, our Terbinafine Hydrochloride provides a reliable benchmark for efficacy. We support global supply chains with timely logistics and regulatory documentation to facilitate smooth market entry for finished products.



