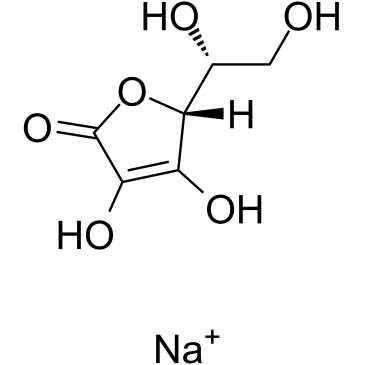
Sodium erythorbate
- CAS No.6381-77-7
- GradeIndustrial / Pharmaceutical
- Availability● In Stock
Premium grade Sodium erythorbate serves as a potent antioxidant and preservative for food and pharmaceutical applications, ensuring product stability and safety.
Request Bulk PricingProduct Technical Details
Product Overview
Sodium erythorbate, chemically known as the sodium salt of D-isoascorbic acid, is a high-performance antioxidant widely utilized across the nutraceutical, food, and pharmaceutical industries. As a stereoisomer of L-ascorbic acid (Vitamin C), it offers exceptional stability and reducing properties without the biological activity of vitamin C, making it ideal for specific preservation tasks. Our manufacturing process ensures high purity levels, meeting stringent international standards for safety and efficacy.
This compound is primarily valued for its ability to prevent oxidation, thereby maintaining the color, flavor, and nutritional integrity of various formulations. It is particularly effective in meat processing, beverage production, and pharmaceutical excipient roles where oxidative degradation must be minimized. Our facility adheres to rigorous quality control protocols to deliver consistent batch-to-batch performance.
Key Specifications
| Parameter | Specification | Typical Result |
|---|---|---|
| Appearance | White or light yellow crystalline powder | White powder |
| Assay (Content) | ≥98.0% | 99.1% |
| pH Value (10% Solution) | 5.5 - 8.0 | 7.4 |
| Specific Rotation | +95.5° to +98.0° | +96.4° |
| Loss on Drying | ≤0.25% | 0.06% |
| Heavy Metals (Pb) | ≤5 mg/kg | ND |
| Arsenic (As) | ≤3 mg/kg | ND |
Industrial Applications
In the food industry, Sodium erythorbate is extensively used as a curing accelerator in meat and poultry products. It helps stabilize color by reducing nitrites to nitric oxide, ensuring the appealing pink hue of cured meats while inhibiting the formation of harmful nitrosamines. Additionally, it serves as a dough conditioner in baking and an antioxidant in beverages to prevent flavor degradation.
Within the pharmaceutical sector, it functions as a critical excipient in formulations susceptible to oxidation. It protects active pharmaceutical ingredients (APIs) from degradation during storage, extending shelf life and maintaining potency. Its high solubility in water facilitates easy integration into liquid formulations and injectables where oxygen scavenging is required.
Quality and Safety
We prioritize safety and compliance in every production run. Our Sodium erythorbate is manufactured under Good Manufacturing Practices (GMP) and is compliant with FCC, USP, and EP standards. Each batch undergoes comprehensive testing for purity, heavy metals, and microbial limits. We provide a Certificate of Analysis (COA) with every shipment to guarantee transparency and quality assurance.
Storage recommendations include keeping the product in a cool, dry environment away from direct sunlight and moisture. Containers should remain tightly sealed to prevent absorption of humidity, which could affect flowability and stability. With proper storage, the product maintains its specified properties for extended periods, ensuring reliability for long-term supply chains.