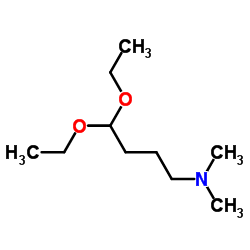
4,4-Diethoxy-N,N-dimethyl-1-butanamine
- CAS No.1116-77-4
- GradeIndustrial / Pharmaceutical
- Availability● In Stock
High purity 4,4-Diethoxy-N,N-dimethyl-1-butanamine serves as a critical pharmaceutical intermediate for the synthesis of migraine treatment APIs.
Request Bulk PricingProduct Technical Details
Product Overview
4,4-Diethoxy-N,N-dimethyl-1-butanamine is a specialized organic compound widely recognized for its pivotal role in pharmaceutical manufacturing. As a key building block, this chemical facilitates the efficient synthesis of complex therapeutic agents, particularly within the class of triptan medications used for migraine management. Our facility ensures that every batch meets rigorous international standards for purity and consistency, supporting reliable downstream production.
The molecular structure features a dimethylamino group protected by a diethoxy acetal functionality. This configuration provides excellent stability during storage and handling while allowing for selective deprotection during specific synthetic transformations. Such chemical versatility makes it an indispensable reagent for medicinal chemists and process engineers focused on developing high-quality active pharmaceutical ingredients.
Key Specifications
| Parameter | Value |
|---|---|
| CAS Number | 1116-77-4 |
| Molecular Formula | C10H23NO2 |
| Molecular Weight | 189.29 g/mol |
| Appearance | Colorless to light-yellowish lucent liquid |
| Purity | ≥98% |
| Boiling Point | 230.7°C at 760 mmHg |
| Density | 0.844 g/cm3 |
| Refractive Index | 1.421 |
Industrial Applications
This compound is primarily utilized as a critical intermediate in the production of several well-known pharmaceutical agents. Its high reactivity and specific structural attributes enable the construction of indole-based derivatives essential for serotonin receptor agonists. Major applications include the synthesis pathways for Zolmitriptan and Sumatriptan, both of which are vital treatments for acute migraine attacks.
By maintaining strict control over impurities and side products, we ensure that our material integrates seamlessly into multi-step synthesis routes. This reduces purification burdens in later stages and enhances the overall yield of the final API. Our clients rely on this consistency to meet regulatory compliance requirements for drug substance manufacturing.
Quality Assurance and Storage
Quality is paramount in pharmaceutical supply chains. Each production lot undergoes comprehensive analysis using advanced chromatographic and spectroscopic methods. A Certificate of Analysis (COA) is provided with every shipment, detailing physical constants and purity metrics. We adhere to Good Manufacturing Practices (GMP) guidelines to guarantee product integrity from synthesis to delivery.
For optimal preservation, the material should be stored in a cool and dry environment within well-closed containers. It is essential to keep the product away from moisture, strong light, and excessive heat to prevent hydrolysis of the acetal group. Standard packaging includes 25kg cardboard drums, though custom solutions are available to meet specific logistical requirements.



