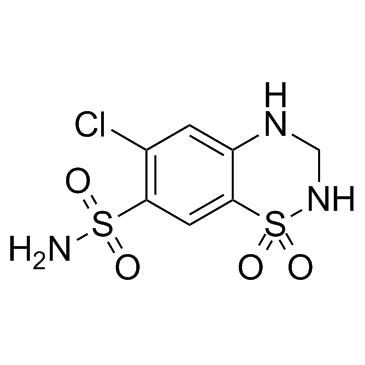
Hydrochlorothiazide
- CAS No.58-93-5
- GradeIndustrial / Pharmaceutical
- Availability● In Stock
High purity Hydrochlorothiazide API compliant with USP standards for pharmaceutical formulations.
Request Bulk PricingProduct Technical Details
Product Overview
Hydrochlorothiazide is a widely recognized pharmaceutical intermediate and active ingredient utilized extensively in the development of cardiovascular and renal therapeutics. As a leading supplier in the global chemical market, we provide this compound with exceptional purity levels suitable for large-scale manufacturing. Our production processes adhere to strict quality management systems, ensuring consistency and reliability for downstream formulation partners.
This compound functions primarily as a diuretic and antihypertensive agent. It is chemically stable and designed to meet the rigorous demands of modern pharmaceutical synthesis. Our facility employs advanced crystallization techniques to achieve the specified particle size and morphology, facilitating optimal processing during tablet compression or capsule filling operations.
Quality Specifications
We maintain rigorous control over every batch to ensure compliance with international pharmacopoeia standards. The following table outlines the critical quality attributes verified through our analytical laboratories. Each parameter is tested using validated methods including HPLC, IR, and UV spectroscopy.
| Items of Analysis | Standard of Analysis | Test Results |
|---|---|---|
| Characters | White or practically white crystalline powder | Conforms |
| Identification | IR, UV | Conforms |
| Loss on drying | ≤0.5% | 0.1% |
| Residue on ignition | ≤0.1% | 0.04% |
| Heavy metals | ≤ 0.001% | Conforms |
| Related compounds | Total other impurities ≤0.9% | 0.5% |
| Assay | 98.0~102.0% | 100.1% |
| Conclusion | Conforms to USP42 | |
Industrial Applications
Our Hydrochlorothiazide is primarily intended for use by licensed pharmaceutical manufacturers. It serves as a key component in the production of antihypertensive medications and diuretic formulations. The high purity profile ensures minimal interference with other active ingredients in combination therapies.
- Suitable for solid dosage forms including tablets and capsules.
- Compatible with various excipients used in cardiovascular drug formulations.
- Supports development of generic equivalents with bioequivalence standards.
- Ideal for research and development in renal therapeutic pathways.
Storage and Handling
To maintain product integrity, proper storage conditions are essential. The material should be kept in a low temperature, ventilated, and dry warehouse environment. It is stable under recommended conditions but should be protected from strong oxidizing agents. Personnel handling this chemical should follow standard safety protocols including the use of appropriate personal protective equipment.
We offer comprehensive documentation including Certificates of Analysis (COA), Method Validation Reports, and Stability Data to support regulatory filings. Our logistics network ensures secure and timely delivery to qualified facilities worldwide.



