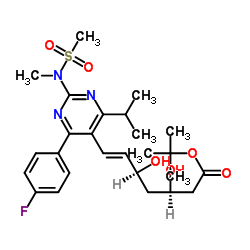
tert-Butyl Rosuvastatin
- CAS No.355806-00-7
- GradeIndustrial / Pharmaceutical
- Availability● In Stock
tert-Butyl Rosuvastatin is a high-purity pharmaceutical intermediate used in the synthesis of Rosuvastatin Calcium. We offer global supply with comprehensive quality documentation.
Request Bulk PricingProduct Technical Details
Product Overview
tert-Butyl Rosuvastatin serves as a pivotal advanced intermediate in the pharmaceutical synthesis pathway of Rosuvastatin Calcium, a widely recognized HMG-CoA reductase inhibitor. This compound features a specific tert-butyl ester protecting group that is essential for maintaining chemical stability during multi-step organic synthesis. Our manufacturing process ensures that each batch meets rigorous international standards for purity and consistency, making it an ideal choice for large-scale API production.
The chemical structure, defined by the molecular formula C26H36FN3O6S, requires precise handling and sophisticated synthesis routes to achieve the desired stereochemistry and functional group integrity. As a key building block, this intermediate facilitates the efficient construction of the final active pharmaceutical ingredient while minimizing impurity profiles.
Key Specifications
We adhere to strict quality control protocols to guarantee product reliability. The following table outlines the critical analytical parameters verified during our internal quality assurance processes.
| Items of Analysis | Standard of Analysis | Test Results |
|---|---|---|
| Appearance | White or almost white crystalline powder | Conform |
| Water | ≤0.5% | 0.3% |
| Related Substances (Individual) | ≤0.2% | 0.08% |
| Related Substances (Total) | ≤1.0% | 0.2% |
| Assay | ≥99.0% | 99.8% |
| Conclusion | Conforms to Factory Standard | |
Industrial Applications
This intermediate is primarily utilized in the medicinal chemistry sector for the production of cardiovascular therapeutics. The tert-butyl ester moiety provides essential protection against unwanted side reactions during the coupling and deprotection stages of synthesis. Pharmaceutical manufacturers rely on this material to streamline their production workflows, ensuring high yields and reduced waste.
Our supply chain supports both pilot-scale development and commercial manufacturing needs. By providing a consistent source of high-quality intermediate, we help partners accelerate their time-to-market for generic and proprietary statin formulations. The material is compatible with standard organic synthesis protocols used in modern API facilities.
Quality Assurance
Quality is the cornerstone of our operation. Every batch of tert-Butyl Rosuvastatin undergoes comprehensive testing using high-performance liquid chromatography (HPLC) and other advanced analytical techniques. We maintain full traceability from raw material sourcing to final packaging.
- Strict adherence to GMP guidelines during production.
- Comprehensive Certificate of Analysis (COA) provided with every shipment.
- Low impurity profiles ensuring safer downstream processing.
- Stable supply chain capable of meeting global demand fluctuations.
Storage and Handling
To maintain optimal stability, this product should be stored in a cool, dry, and well-ventilated area away from direct sunlight and moisture. Containers must be kept tightly closed when not in use to prevent hydrolysis or contamination. Proper personal protective equipment (PPE) is recommended during handling to ensure safety in accordance with standard laboratory and industrial hygiene practices. For detailed safety data, please refer to the provided SDS documentation.



